No products in the cart.
6-APDB crystal rocks
€16.95
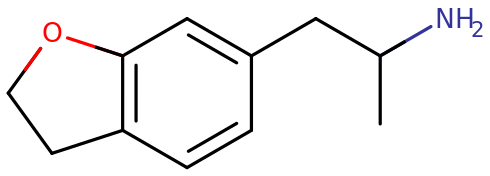
6-APDB, known more formally as 2,3-dihydro-α-methyl-6-benzofuranethanamine, is an analog of the psychedelic drugs MDA and 6-APB. This compound fully substitutes for the entactogen MBDB in animal drug-discrimination studies and blocks the reuptake of serotonin by rat synaptosomes. This product is intended for forensic and research applications.
Purity: 97.5%
| 1 Gram | €16.95 |
| 3 Grams | €29.95 |
| 5 Grams | €39.95/td> |
| 10 Grams | €69.95 |
| 25 Grams | €129.95 |
6-APDB crystal rocks – More information
6-APDB (also known as 6-(2-aminopropyl)-2,3-dihydrobenzofuran or 4-Desoxy-MDA) is a stimulant and entactogenic research chemical of the phenethylamine and benzofuran classes. It is a closely related synthetic analogue of MDA and 6-APB and broadly shares the characteristics of serotonin-selective triple monoamine releasers and reuptake inhibitors associated with other entactogenic or empathogenic compounds.
6-APDB was first synthesized and studied along with 5-APDB in 1993 by David E. Nichols as a potential non-neurotoxic alternative to MDMA. It did not come into popular recreational use until over a decade later, where it briefly entered the rave scene and global research chemicals market, in particular the “legal highs” market in the U.K., before its sale and import were subsequently banned.
Because 6-APDB and other substituted benzofurans have not been explicitly outlawed in some countries, they are often technically legal, contributing to their popularity as a substitute or replacement for serotonergic entactogens like MDMA or MDA, and are typically distributed through the online research chemicals grey market.
clinical data
| Common names | 6-APDB |
| Substitutive name | 6-(2-aminopropyl)-2,3-dihydrobenzofuran |
| Systematic name | 1-(2,3-dihydrobenzofuran-6-yl)propan-2-amine |
| Psychoactive class | Entactogen / Stimulant |
| Chemical class | Amphetamine / Benzofuran |
6-APDB dosage table
| Threshold | 20 – 30 mg |
| Light | 30 – 70 mg |
| Common | 70 – 100 mg |
| Strong | 100 – 130 mg |
| Heavy | 130 mg + |
6-APDB effect progress
| Total | 6 – 8 hours |
| Offset | 2 – 3 hours |
Chemistry
6-APDB, also known as 6-(2-aminopropyl)-2,3-dihydrobenzofuran, is a synthetic molecule of the benzofuran family. Molecules of this class contain a phenethylamine core bound to an amino (NH2) group through an ethyl chain with an additional methyl substitution at Rα.
6-APDB does not contain a methyl substitution on RN, a motif which it shares with MDA. It is an analogue of MDA where the heterocyclic 4-position oxygen from the 3,4-methylenedioxy ring has been replaced with a methylene bridge. 6-APDB shares this furan ring with 5-APDB, 5-MAPDB and 6-MAPDB.
Toxicity
Due to only having a short history of human use, the toxicity and harm potential is not exactly known. Due to its similarity to MDMA, it is likely that the administration of repeated or high dosages of 6-APDB can be neurotoxic and cardiotoxic in some form.
The exact toxic dosage is unknown. It is strongly recommended that one use harm reduction practices when using this drug.
Short-term health concerns
Short-term physical health risks of 6-APDB consumption include dehydration, insomnia, and hyperthermia. Continuous activity without sufficient rest or rehydration may cause body temperature to rise to dangerous levels, and loss of fluid via excessive perspiration puts the body at further risk as the stimulatory and euphoric qualities of the drug may render the user oblivious to their energy expenditure for quite some time. Diuretics such as alcohol may exacerbate these risks further, although this is known to be more of a problem for MDMA than it is 6-APDB.
Although it has not been formally studied, like with MDMA, small changes in ambient temperature may cause large changes in 6-APDB-induced serotonin neurotoxicity and core body temperature in the rat.
Long-term health concerns
The neurotoxicity of 6-APDB is controversial. It was specifically designed to be less neurotoxic than MDA or MDMA by circumventing the production of certain metabolic byproducts thought to underlie their toxicity (specifically alpha-methyl-dopamine). Although it is likely to be physically safe to try in a responsible context, it is completely possible that the administration of repeated or high dosages of 6-APDB could result in neurotoxic effects in some form, possibly manifesting as deficits in cognitive, affective and psychomotor function.
As with MDMA, long-term heavy use of 6-APDB is likely cardiotoxic and thought to lead to valvulopathy through its actions on the 5-HT2B receptor.
Tolerance
As with other stimulants, the chronic use of 6-APDB can be considered moderately addictive with a high potential for abuse and is capable of causing psychological dependence among certain users. When addiction has developed, cravings and withdrawal effects may occur if a person suddenly stops their usage.
As a potent releaser of serotonin, tolerance builds quickly with prolonged and repeated use to the point that the drug eventually loses any positive effects and instead leaves the user in an uncomfortable state of anxious stimulation and dysphoria. This results in users having to administer increasingly large doses to achieve the same effects. After that, it takes about 21-30 days for the tolerance to be reduced to half and 2-3 months to be back at baseline (in the absence of further consumption). 6-APDB presents cross-tolerance with all dopaminergic stimulants, meaning that after the consumption of 6-APDB all stimulants will have a reduced effect.
Serotonin syndrome risk
Combinations with the following substances can cause dangerously high serotonin levels. Serotonin syndrome requires immediate medical attention and can be fatal if left untreated.
There is an increased risk of serotonin syndrome when 6-APDB is taken with many antidepressants, particularly selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs). Additionally, if 6-APDB is taken with SSRIs and SNRIs, the 6-APDB will be significantly less powerful or may have no distinguishable effects at all.
| amount | 1 Gram, 3 Grams, 5 Grams, 10 Grams, 25 Grams |
|---|
1 review for 6-APDB crystal rocks
Only logged in customers who have purchased this product may leave a review.
Related products
Entactogens
€11.95
Entactogens
€11.95
Stimulant
€10.95
Stimulant
€13.95
Entactogens
Rated 4.50 out of 5
€11.95Stimulant
€11.95
Entactogens
€11.95
Stimulant
€13.95




















JoeT42 –
delayed, in return got a free sample pack so im hapy