No products in the cart.
LSZ blotters 150 µg
€13.95
LSZ likely acts as a 5-HT2A partial agonist. The psychedelic effects are believed to come from LSZ’s efficacy at the 5-HT2A receptors. However, the role of these interactions and how they result in the psychedelic experience continues to remain elusive.
Purity: 96.5%
| 5 Blotters | €13.95 |
| 15 Blotters | €35.95 |
| 25 Blotters | €55.95 |
| 50 Blotters | €91.95 |
| 125 Blotters | €178.95 |
LSZ blotters 150 µg – More information
Lysergic acid 2,4-dimethylazetidide (also known as λ, Lambda, and LSZ) is a synthetic psychedelic of the lysergamide chemical class which produces LSD-like psychedelic effects when administered.
In the 2000s, a team led by David E. Nichols at Purdue University set to develop a rigid analog of LSD with the diethylamide group constrained into an azetidine ring in order to map the binding site at the 5-HT2A receptor.
LSZ has little to no history of human usage prior to 2012 when it appeared on some research chemical markets in the UK. LSZ later gained international popularity through a small cluster of mail-order novel psychedelic shops that appeared in 2012. There have also been several unconfirmed reports of LSZ being synthesized in illicit laboratories and distributed on blotter paper or in liquid solution under names such as “Diazedine” and “λ” (or “Lambda”).
LSZ is not considered to be addictive or physiologically toxic. Nevertheless, adverse psychological reactions such as severe anxiety, paranoia and psychosis are always possible, particularly among those predisposed to mental illness. It is highly advised to use harm reduction practices if using this substance.
clinical data
| Common names | LSZ, LA-SS-Az, Diazedine, Lambda |
| Substitutive name | Lysergic acid 2,4-dimethylazetidide |
| Systematic name | (8β)-8-{[(2S,4S)-2,4-Dimethylazetidin-1-yl]carbonyl}-6-methyl-9,10-didehydroergoline |
| Psychoactive class | Psychedelic |
| Chemical class | Lysergamide |
LSZ dosage table
| Threshold | 50 – 100 µg |
| Light | 100 – 150 µg |
| Common | 150 – 300 µg |
| Strong | 300 – 400 µg |
| Heavy | 400 µg + |
LSZ effect progress
| Total | 6 – 10 hours |
| Offset | 2 – 3 hours |
| After effects | 6 – 24 hours |
Chemistry
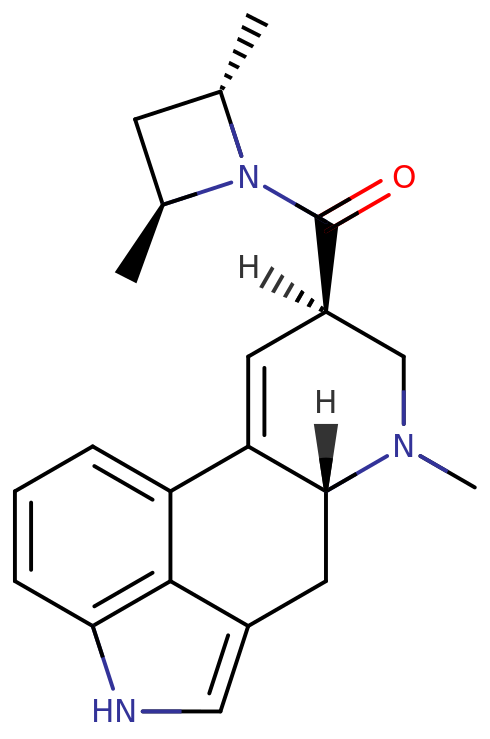
LSZ, or d-lysergic acid 2,4-dimethylazetidide, is a semi-synthethic alkaloid of the lysergamide famiy. It contains a core structure of lysergic acid with an amine functional group bound to RN of the chemical structure. This core polycyclic structure is an indole derivative, and has tryptamine and phenethylamine groups embedded within it.
The structure contains a bicyclic hexahydroindole fused to a bicyclic quinoline group (lysergic acid). At carbon 8 of the quinoline, an amide group is bound. Additionally, the substitutions of the terminal nitrogen atom of the amide group form a 2,4-dimethylazetidide group. LSZ is additionally substituted at carbon 6 with a methyl group.
There are three possible stereoisomers around the azetidine ring with the (S,S)-(+) isomer being the most active. It is slightly more potent than LSD itself in drug discrimination tests using trained rats.
Toxicity
The toxicity and long-term health effects of recreational LSZ do not seem to have been studied in any scientific context and the exact toxic dose is unknown. This is because LSZ is a research chemical with very little history of human usage. Anecdotal evidence from people within the psychonaut community who have tried LSZ suggests that there are no negative health effects attributed to simply trying the drug by itself at low to moderate doses and using it very sparingly (but nothing can be completely guaranteed). Independent research should always be done to ensure that a combination of two or more substances is safe before consumption.
It is strongly recommended that one use harm reduction practices when using this substance.
Tolerance
LSZ is not habit-forming and the desire to use it can actually decrease with use. It is most often self-regulating.
Tolerance to the effects of LSZ are built almost immediately after ingestion. After that, it takes about 3 days for the tolerance to be reduced to half and 7 days to be back at baseline (in the absence of further consumption). LSZ presents cross-tolerance with all psychedelics, meaning that all psychedelics will have a reduced effect after the consumption of LSZ.
References
| amount | 5 Blotters, 15 Blotters, 25 Blotters, 50 Blotters, 125 Blotters |
|---|
Only logged in customers who have purchased this product may leave a review.
Related products
Psychedelic
€10.95
Psychedelic
€10.95
Entactogens
Rated 5.00 out of 5
€11.95Psychedelic
€10.95
Psychedelic
€11.95
Psychedelic
€10.95
Psychedelic
€13.95
Psychedelic
€11.95






















Reviews
There are no reviews yet.