No products in the cart.
Theacrine crystal rocks
€10.95
Theacrine is an analytical reference standard categorized as a stimulant. It also has analgesic properties. This product is intended for research and forensic applications.
Purity: 98.0%
| 1 Gram | €10.95 |
| 3 Grams | €27.95 |
| 5 Grams | €39.95 |
| 10 Grams | €74.95 |
| 25 Grams | €159.95 |
Theacrine crystal rocks – More information
Theacrine (also known as Teacrine) is a nootropic stimulant and a structural analog of caffeine, appearing to be synthesized from caffeine in some plants. It is a bitter, white crystalline purine alkaloid that increases activity in the brain and induces temporary improvements including enhanced alertness, wakefulness, and stimulation. Relative to caffeine, it displays about 2/3rds the potency in terms of the stimulation it produces.
The mechanisms of theacrine largely parallel those of caffeine in that while it seems to have a stimulatory effect in rodents, this only occurs at a higher dose (and the exact oral dose where it peaks with theacrine is not known). Similar to caffeine, it produces a sedative effect at relatively low doses, but where this sedative effect with caffeine is at an impractically low dose with theacrine it is the dose normally consumed by tea; this may underlie why Kucha tea tends to be recommended for relaxation more than stimulation.
clinical data
| Common names | Theacrine, Temurin, Temorine |
| Substitutive name | 1,3,7,9-Tetramethyluric acid |
| Systematic name | 1,3,7,9-Tetramethylpurine-2,6,8-trione |
| Psychoactive class | Stimulant |
| Chemical class | Purine alkaloid |
Theacrine dosage table
| Threshold | 25 mg |
| Light | 50 – 100 mg |
| Common | 25 – 100 mg |
| Strong | 150 – 300 mg |
| Heavy | 300 mg + |
Theacrine effect progress
| Total | 1 – 3 hours |
Chemistry
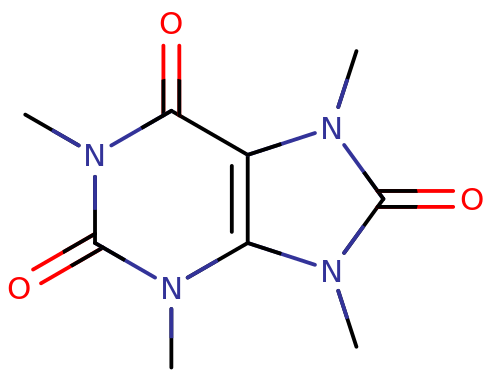
Theacrine’s chemical structure is structurally similar to that of caffeine. It is a synthetic alkaloid with a substituted xanthine core. Xanthine is a substituted purine, which contains two fused rings, a pyrimidine and imidazole. Pryimidine is a 6 membered ring with nitrogen constituents at R1 and R3; imidazole is a 5 membered ring with nitrogen substituents at R1 and R3. Xanthine contains oxygen groups double-bonded to R2 and R6. Like caffeine, it contains additional methyl substitutions at R1, R3, and R7 of its structure, bound to the open nitrogen groups of the xanthine skeleton. It is an achiral aromatic compound.
Theacrine’s chemical name is 1,3,7,9-tetramethyluric acid; in comparison, the chemical name of caffeine is 1,3,7-trimethylxanthine, with the only difference in structure being an additional methyl group on the 9-carbon and an additional ketone group, which changes caffeine’s xanthine into a uric acid moiety.
Toxicity
Theacrine is not known to cause brain damage, and has an extremely low toxicity relative to dose. There are relatively few physical side effects associated with theacrine exposure. Various studies have shown that in reasonable doses in a careful context, it presents no negative cognitive, psychiatric or toxic physical consequences of any sort.
It is strongly recommended that one use harm reduction practices when using this drug.
Tolerance
As with other stimulants, the chronic use of theacrine can be considered moderately addictive with a high potential for abuse and is capable of causing psychological dependence among certain users. When addiction has developed, cravings and withdrawal effects may occur if a person suddenly stops their usage.
Tolerance to many of the effects of theacrine develops with prolonged and repeated use. This results in users having to administer increasingly large doses to achieve the same effects. After that, it takes about 3 – 7 days for the tolerance to be reduced to half and 1 – 2 weeks to be back at baseline (in the absence of further consumption). It should be noted, however, that tolerance buildup does not occur as rapidly as with that of caffeine.
| amount | 1 Gram, 3 Grams, 5 Grams, 10 Grams, 25 Grams |
|---|
Only logged in customers who have purchased this product may leave a review.
Related products
Stimulant
€10.95
Psychedelic
Rated 3.00 out of 5
€11.95Stimulant
€10.95
Entactogens
€11.95
Entactogens
Rated 4.50 out of 5
€11.95Stimulant
€10.95
Stimulant
€13.95
Stimulant
€10.95



















Reviews
There are no reviews yet.