No products in the cart.
4F-EPH crystal rocks
€13.95
4F-EPH is believed to act as a higher efficiency dopamine reuptake inhibitor than the closely related methylphenidate, meaning that it effectively boosts the levels of dopamine neurotransmitters in the brain by binding to and partially blocking the transporter proteins that normally remove those monoamines from the synaptic cleft. This allows dopamine to accumulate within the certain focalized regions of the brain, resulting in stimulating and euphoric effects.
Purity: ≥97.0%
| 1 Gram | €13.95 |
| 3 Grams | €35.95 |
| 5 Grams | €55.95 |
| 10 Grams | €91.95 |
| 25 Grams | €178.95 |
4F-EPH crystal rocks – More information
4-Fluoroethylphenidate (commonly known as 4F-EPH) is a novel synthetic stimulant of the substituted phenethylamine and piperidine classes that produces long-lasting euphoric and stimulating effects associated with potent monoamine reuptake inhibitors when administered. It is a closely related structural analog of the commonly prescribed ADHD drug methylphenidate (known by the brand-names Ritalin and Concerta) as well as a designer drug analog ethylphenidate. Based on its similarities to other memebers of this class, it is speculated to exert its activity as some form of double or triple monoamine reuptake inhibitor.
Like other members of the substituted phenidate family, anecdotal reports suggest that 4F-EPH can be corrosive to the nasal cavities, albeit not to the degree of ethylphenidate.
4F-EPH has little to no history of recreational use and has yet to be documented being sold on the streets. It was initially developed as a replacement and successor for compounds like ethylphenidate, which became illegal in the United Kingdom on April 2015, and later 4F-MPH. In 2016, it became made available for sale on the online gray market as a research chemical.
Due to its potent, long-lasting stimulant effect, likely habit-forming properties as well as unknown short and long-term toxicity profile, it is strongly recommended that one use proper harm reduction practices if choosing to use this substance.
clinical data
| Common names | 4F-EPH, 4FEPH |
| Substitutive name | 4-Fluoroethylphenidate |
| Systematic name | Ethyl 2-(4-fluorophenyl)-2-(piperidin-2-yl)acetate |
| Psychoactive class | Stimulant |
| Chemical class | Phenidate / Piperidine |
4F-EPH dosage table
| Threshold | < 5 mg |
| Light | 5 – 10 mg |
| Common | 10 – 15 mg |
| Strong | 15 – 30 mg |
| Heavy | 30 mg + |
4F-EPH effect progress
| Total | 5 – 6 hours |
| After effects | 4 – 12 hours |
4F-EPH – Chemistry
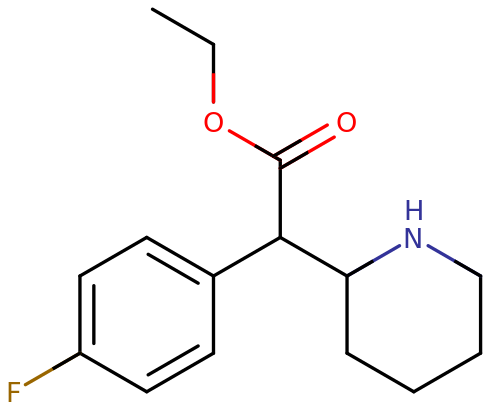
4F-EPH, or 4-fluoroethylphenidate, is a synthetic molecule of the substituted phenethylamine class. It contains a phenethylamine core featuring a phenyl ring bound to an amino -NH2 group through an ethyl chain. It is structurally similar to amphetamine, featuring a substitution at Rα which is incorporated into a piperidine ring ending at the terminal amine of the phenethylamine chain. It contains an ethyl acetate bound to R2 of its structure and is fluorinated at R4 of its phenyl ring.
4F-EPH is the 4-substituted flourine derivative of ethylphenidate, and is structurally different to methylphenidate by elongation of the carbon chain and the addition of a fluorine group. Ethyl- regards the side chain of two carbon atoms, phen- indicates the phenyl ring, id- is contracted from a piperidine ring, and -ate indicates the acetate group containing the oxygens. 4F-EPH is a chiral compound, presumably produced as a racemic mixture.
Toxicity
The toxicity and long-term health effects of recreational 4F-EPH use do not seem to have been studied in any scientific context and the exact toxic dosage is unknown. This is because 4F-EPH is a research chemical with very little history of human usage. Anecdotal evidence from people who have tried 4F-EPH suggests that there are no negative health effects attributed to simply trying the drug by itself at low to moderate doses and using it very sparingly (but nothing can be completely guaranteed).
It is strongly recommended that one use harm reduction practices when using this drug.
4F-EPH – Tolerance
As with other stimulants, the chronic use of 4F-EPH can be considered moderately addictive with a high potential for abuse and is capable of causing psychological dependence among certain users. When addiction has developed, cravings and withdrawal effects may occur if a person suddenly stops their usage.
Tolerance to many of the effects of 4F-EPH develops with prolonged and repeated use. This results in users having to administer increasingly large doses to achieve the same effects. After that, it takes about 3 – 7 days for the tolerance to be reduced to half and 1 – 2 weeks to be back at baseline (in the absence of further consumption). 4F-EPH presents cross-tolerance with all dopaminergic stimulants, meaning that after the consumption of 4F-EPH all stimulants will have a reduced effect.
| amount | 1 Gram, 3 Grams, 5 Grams, 10 Grams, 25 Grams |
|---|
Only logged in customers who have purchased this product may leave a review.
Related products
Stimulant
€10.95
Stimulant
€11.95
Sale!
New
Antipsychotic
Stimulant
€13.95
Entactogens
€11.95
Entactogens
Rated 4.50 out of 5
€11.95Stimulant
€13.95
Stimulant
€13.95





















Reviews
There are no reviews yet.